Zydus Cadila gets USFDA nod to market Potassium Chloride capsules
The company has more than 254 approvals and has so far filed over 350 abbreviated new drug applications (ANDAs) since it commenced filings in 2003-04.
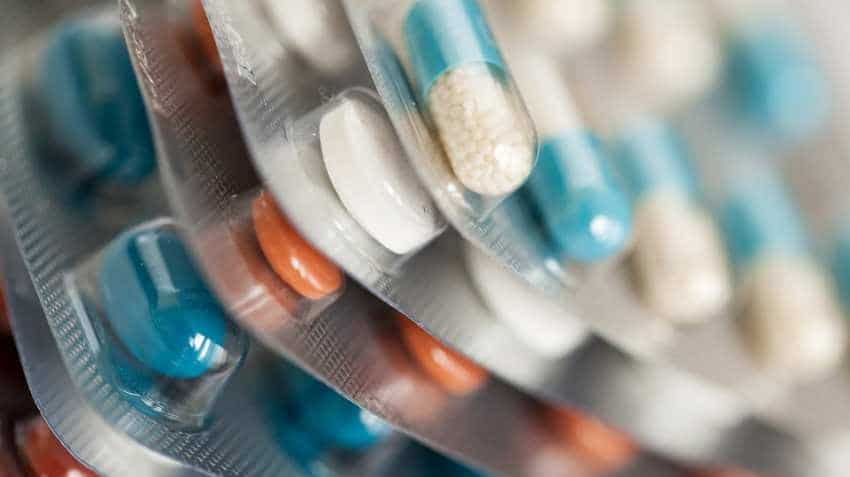
This medication is a mineral supplement used to treat or prevent low amounts of potassium in the blood. Photo: Pixabaypti





 Zydus gets US regulator's PAS for generic version of Mycophenolate Mofetil for injection
Zydus gets US regulator's PAS for generic version of Mycophenolate Mofetil for injection Zydus gets approval to conduct Phase II clinical study in patients with CAPS in Australia
Zydus gets approval to conduct Phase II clinical study in patients with CAPS in Australia Zydus Cadila's Covid-19 vaccine ZyCoV-D to be used in these seven states initially
Zydus Cadila's Covid-19 vaccine ZyCoV-D to be used in these seven states initially Zydus Cadila gets USFDA nod to market generic cancer drug in US
Zydus Cadila gets USFDA nod to market generic cancer drug in US